The monohydrate loses its water at 100-110 ☌. 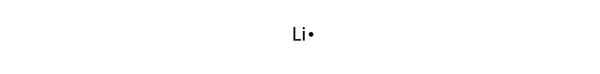
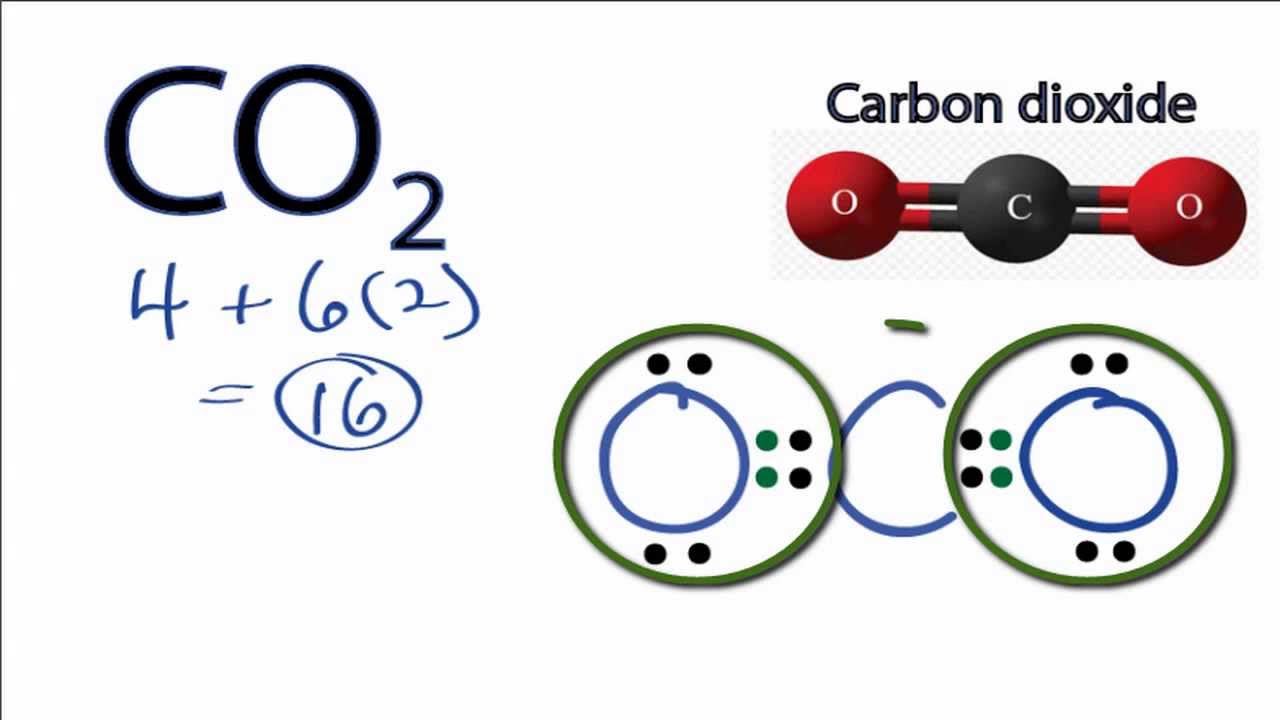
1 gram of anhydrous lithium hydroxide can remove 450 cm 3 of carbon dioxide gas. The later, anhydrous hydroxide is preferred for its lower mass and lesser water production for respirator systems in spacecraft. Lithium hydroxide is used in breathing gas purification systems for spacecraft (Lithium hydroxide canisters in the LEM were lifelines for the Apollo 13 astronauts), submarines, and rebreathers to remove carbon dioxide from exhaled gas by producing lithium carbonate and water: Write a net ionic equation for this reaction. Li 2CO 3 + Ca(OH) 2 → LiOH + CaCO 3 Reactions When an aqueous solution of hydroiodic acid, HI (aq), and an aqueous solution of ammonium carbonate, (NH4)2CO3 (aq), are combined, the gas carbon dioxide, CO2(g), is released from solution. Industrially, lithium hydroxide is produced in a metathesis reaction between lithium carbonate and calcium hydroxide: The reaction is as follows:Ģ Li + 2 H 2O → 2 LiOH + H 2 Li 2O + H 2O → 2 LiOHīecause lithium reacts rapidly (but not violently) with water, lithium batteries should be kept away from water. Likewise, the chemical finds application in the glass-making industry. The balanced chemical equation is given by 2 LiOH(s). It is also usable in the health industry for the treatment of specific diseases. Lithium hydroxide can be produced by dissolving lithium or lithium oxide in water. The lithium hydroxide reacts with carbon dioxide to form solid lithium carbonate (Li CO ) and liquid water. Disclosed is a carbon dioxide gas absorbent containing lithium silicate reacting with a carbon dioxide gas to form lithium carbonate and represented by the. annual CO 2 total output emission rate lb/MWh, year 2019 data) Notes: This calculation does not include any greenhouse gases other than CO 2. It is also used in ceramics, manufacturing other lithium compounds, and esterification specially for lithium stearate (which is used as a general purpose lubricating grease due to its high resistance to water and is useful at both high and low temperatures). 884.2 lbs CO2/MWh × 1 metric ton/2,204.6 lbs × 1/ (1-0.073) MWh delivered/MWh generated × 1 MWh/1,000 kWh × 4.33 × 10-4 metric tons CO2/kWh (eGRID, U.S. Addition of hydrochloric acid (HCl) produces lithium chloride, which is the compound used to produce lithium metal by electrolysis. It is used as a heat transfer medium, as a storage- battery electrolyte, and as a catalyst for polymerization. The major commercial form is lithium carbonate, Li 2 CO 3, produced from ores or brines by a number of different processes. The reaction between limewater, which is a solution of calcium hydroxide, 'Ca(OH)'2, and carbon dioxide will result in the formation of an insoluble solid called calcium carbonate, 'CaCO'''3. Lithium hydroxide is used in carbon dioxide scrubbers for purification of gases and air. Carbon dioxide reacts with limewater to form calcium carbonate, which precipitates out of the solution. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
